Product Name:Liraglutide
Cas No.204656-20-2
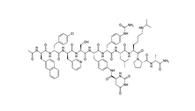
SequenceH-His-Ala-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Val-Ser-Ser-Tyr-Leu-Glu-Gly-Gln-Ala-Ala-Lys(N-ε-(Nα-Palmitoyl-L-γ-glutamyl))-Glu-Phe-Ile-Ala-Trp-Leu-Val-Arg-Gly-Arg-Gly-OH
Molecular FormulaC172H265N43O51
Molar Mass3751.20
The Maximum Unspecified Impurity ≤0.2%
Storage Temperature 5℃±3℃
Packing Size100G/Bottle or at customers reqirement.
Liraglutide, an analog of human GLP-1, and acts as a GLP-1 receptor agonist. The recombinant peptide precursor of liraglutide, produced by its expression in Saccharomyces cerevisiae, has been engineered to be 97% homologous to native human GLP-1 by substituting R for K at position 34. Liraglutide was designed by attaching a C-16 fatty acid (palmitic acid) with a glutamic acid spacer on the remaining lysine residue at position 26 of the peptide precursor.
Indication
For use in/treatment of diabetes mellitus type 2.
Pharmacodynamics
Liraglutide is a once-daily GLP-1 derivative for the treatment of type 2 diabetes. GLP-1, in its natural form, is short-lived in the body (the half-life after subcutaneous injection is approximately one hour), so it is not very useful as a therapeutic agent. However, liraglutide has a half-life after subcutaneous injection of 11â€"15 hours, making it suitable for once-daily dosing. The prolonged action of liraglutide is achieved by attaching a fatty acid molecule at one position of the GLP-1 molecule, enabling it to bind to albumin within the subcutaneous tissue and bloodstream. The active GLP-1 is then released from albumin at a slow, consistent rate. Binding with albumin also results in slower degradation and reduced elimination of liraglutide from the circulation by the kidneys compared to GLP-1.
Mechanism of Action
Liraglutide is an acylated GLP-1 (Glucagon-Like Peptide-1) receptor agonist. Liraglutide upregulates intracellular cAMP resulting in the release of insulin given elevated blood glucose concentrations. Glucagon secretion is also decreased in a glucose-dependent fashion by liraglutide.
Metabolism
During the initial 24 hours following administration of a single [3H]-liraglutide dose to healthy subjects, the major component in plasma was intact liraglutide. Liraglutide is endogenously metabolized in a similar manner to large proteins without a specific organ as a major route of elimination.
| Liraglutide sequence: H-His-Ala-Glu-Gly-Thr-Phe-Thr-Ser-Asp-Val-Ser-Ser-Tyr-Leu-Glu-Gly-Gln-Ala-Ala-Lys(γ-Glu-palmitoyl)-Glu-Phe-Ile-Ala-Trp-Leu-Val-Arg-Gly-Arg-Gly-OH |
|
| molecular weight: 3751.20 g/mol |
molecular formula: C172H265N43O51 |
| CAS No.: [204656-20-2] |
Half-Life: approx. 13 hrs |
|
|
|
Absorption
Maximum concentrations of liraglutide are achieved at 8-12 hours after dose. The mean peak and total exposures of liraglutide were 35 ng/mL and 960 ng·h/mL, respectively, for a subQ single dose of 0.6 mg. After subcutaneous single-dose administrations, Cmax and AUC of liraglutide increased proportionally over the therapeutic dose range of 0.6 mg to 1.8 mg. At 1.8 mg Victoza, the average steady-state concentration of liraglutide over 24 hours was approximately 128 ng/mL AUC0-∞ was equivalent between the upper arm and abdomen, and between upper arm and thigh. AUC0-∞ from thigh was 22% lower than that from the abdomen. The absolute bioavailability of liraglutide following subcutaneous administration is approximately 55%.

- Certificates

- INSTRUMENTS



- EQUIPMENTS

Hot Tags: liraglutide, China liraglutide manufacturers, factory, Dipeptide-2, Menthol, Sodium Ascorbate, BRONOPOL, 1 R 3R Methyl 1Benzo d 1 3 diozol 5 yl 2 2 chloroacetyl 2 3 4 9 tetra Hydro 1H pyrido 3 4 b indole 3 carboxylate, L-Carnitine-L-Tartrate